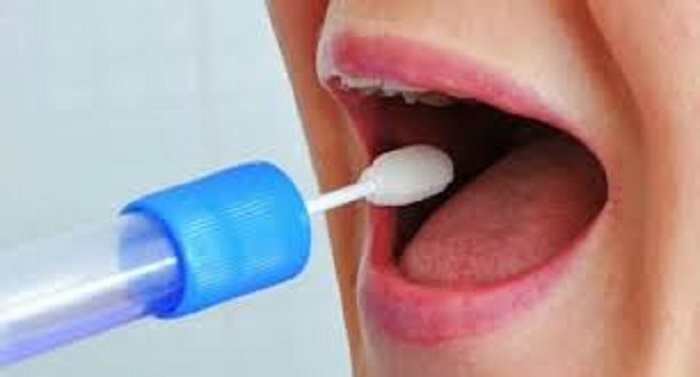
The US Food and Drug Administration on Friday issued an emergency-use authorization for the first at-home Covid-19 test that uses saliva samples, the agency said in a news release.
With the test, people can collect their own saliva at home and send their saliva samples to a lab for results. Testing for Covid-19 so far has usually involved nose or throat swab samples.
Rutgers University's RUCDR Infinite Biologics lab received an amended emergency authorization late Thursday. In April, Rutgers University announced that the FDA authorized the saliva test that it developed with other groups for "emergency use" for diagnosing Covid-19.
"What's new and next is expanding access to testing for people," Andrew Brooks, chief operating officer and director of technology development at the RUCDR Infinite Biologics lab, told CNN.
"If people are committed to do self-collection and can facilitate that collection at home, certainly with a prescription under medical care, we can get to those that are quarantined, don't have the means for transportation or are too scared to go outside," Brooks said. "So they get the test in the mail or from a distribution center."